Research Group Dalpke
Group Leader: Prof. Dr. med Alexander Dalpke
RESEARCH INTERESTS
The focus of my research is infection and immunity. Current work centers on two topics:
1. We are studying the recognition of microbial nucleic acids by pattern recognition receptors. We try to elucidate principles that enable Toll-like receptors to differentiate foreign, microbial from self nucleic acids. Specifically, we address the importance of RNA modifications and RNA processing on immune stimulation. We are also interested in longterm effects of bacterial encounter on innate immune cells.
2. A recent project area studies polymicrobial infections by means of next generation sequencing. We focus on patients suffering from cystic fibrosis (CF) but also use the technique to explore microbiome changes in other diseases. In CF we aim to analyze microbiome alterations in early childhood as well as during exacerbation and focus on interactions of commensal and pathogenic bacteria.
Funding: DFG Da592-7, -12, TRR319, Mukoviszidose e.V., GILEAD, Becton Dickinson
Immunostimulation by nucleic acids
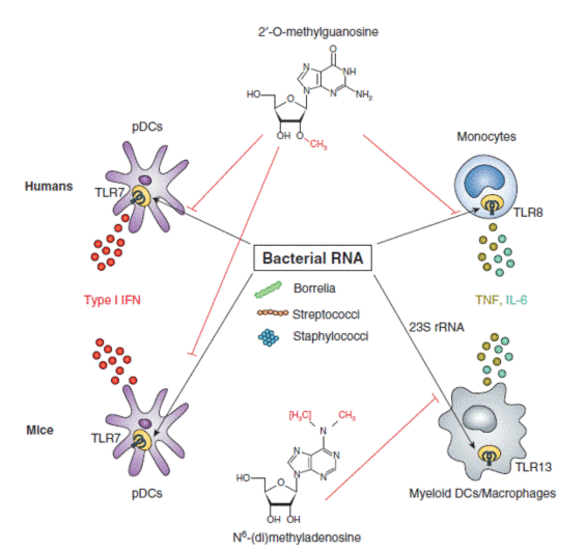
Cells of the innate immune system recognize conserved microbial structures, so called pathogen-associated molecular patterns (PAMPs) by pattern recognition receptors. Among these receptors Toll-like receptors (TLR) 3, -7, -8, -9 and -13 recognize nucleic acids of viral, bacterial or synthetic origin. However, the exact mechanisms of nucleic acid recognition and the differentiation between foreign (dangerous) and self (usually harmless) are still only known partially. Work of our own could show that bacterial RNA represents a PAMP recognized by various immune cells. We could also show that former orphan TLR13 is a murine receptor for bacterial RNA. Posttranscriptional RNA modifications play a decisive role for self/non-self discrimination. We identified 2’O-methylation as a modification that inhibits TLR7 and 8 stimulation. Recent research indicates that also RNA processing is a prerequisite and critical for RNA-TLR interaction; we study how RNA modifications and RNA degradation act together.
Selected publications:
Hafner A, Kolbe U, Freund I, Castiglia V, Kovarik P, Poth P, Herster F, Weigand MA, Weber AN, Dalpke AH and Eigenbrod T (2019): Crucial role of nucleic acid sensing via endosomal Toll-like receptors for the defense of Streptococcus pyogenes in vitro and in vivo. Front Immunol, 10:198.doi: 10.3389/fimmu.2019.00198
Keller P, Freund I, Marchand V, Bec G, Huang R, Motorin Y, Eigenbrod T, Dalpke A, Helm M (2018): Double methylation of tRNA-U54 to 2'-O-methylthymidine (Tm) synergistically decreases immune response by Toll-like receptor 7. Nucleic Acids Res. 46(18):9764-9775
Eigenbrod T and Dalpke AH (2015): Bacterial RNA: An Underestimated Stimulus for Innate Immune Responses. J Immunol 195(2): 411-8
Eigenbrod T, Pelka K, Latz E, Kreikemeyer B and Dalpke AH (2015): TLR8 Senses Bacterial RNA in Human Monocytes and Plays a Nonredundant Role for Recognition of Streptococcus pyogenes. J Immunol 195(3):1092-9
Hidmark, A., A. von Saint Paul, A.H. Dalpke (2012): Cutting Edge: TLR13 is a receptor for bacterial RNA. J Immunol 189(6): 2717-2721
Gehrig, S., M.E. Eberle,F. Botschen, K. Rimbach, F. Eberle, T. Eigenbrod, S. Kaiser, W.M. Holmes, V.A. Erdmann, M. Sprinzl, G. Bec, G. Keith, A.H. Dalpke*, M. Helm* (2012): Identification of modifications in microbial, native tRNA that suppress immunostimulatory activity. J Exp Med 209(2): 225-233 *equal contribution
Microbiome analysis in cystic fibrosis
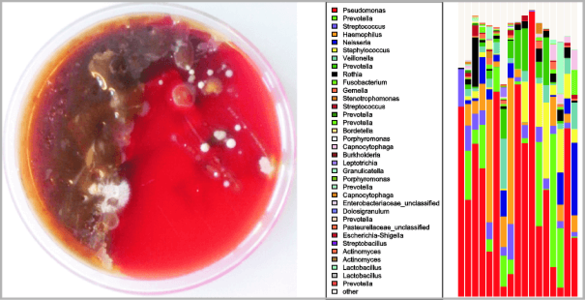
Interactions between bacteria and their host represent a full continuum from pathogenicity to mutualism. From an evolutionary perspective, host-bacteria relationships are no longer considered a two-component ecological system but rather a complex interactive network. Cystic fibrosis (CF) is characterized by defective mucociliary clearance associated with polymicrobial chronic airway infections. Those infections, leading to persistent inflammation and periodic episodes of acute pulmonary exacerbation, contribute to an irreversible decline in CF lung function. However, the link between bacterial infections and decline in CF lung function is not yet understood. We study CF lung microbiome aiming to answer different questions about CF microbiome resilience, antibiotic treatment effect and possible dysbiosis induced by exacerbation. In a recent project we address the hypothesis that certain airway commensal bacteria might interfere with CF pathogens and thus exert rather beneficial effects. This work focuses on interactions of commensal bacteria with Pseudomonas aeruginosa within in vitro systems as well as microbiome and metagenomics studies from patients
Selected publications
Metzger M, Gräber S, Stahl M, Sommerburg O, Mall M, Dalpke A and Boutin S (2021): A volatile and dynamic longitudinal microbiome is associated with less reduction in lung function in adolescents with cystic fibrosis. Front Cell Infect Microbiol, accepted for publication
Frey DL*, Boutin S*, Dittrich S*, Graeber SY, Stahl M, Wege S, Herth F, Sommerburg O, Schultz C, Mall M, Dalpke A (2021): Relationship between airway dysbiosis, inflammation and lung function in adults with cystic fibrosis. J Cyst Fibr, S1569-1993(20):30954-1
Kolbe U, Yi B, Poth T, Saunders A, Boutin S, Dalpke A (2020): Early cytokine induction upon Pseudomonas aeruginosa infection in murine precision cut lung slices depends on sensing of bacterial viability. Front Immunol, 11: 598636. doi: 10.3389/fimmu.2020.598636
Sörensen M, Kantorek J, Byrnes L, Boutin S, Mall MA, Lasitschka F, Zabeck H, Nguyen D and Dalpke AH (2020): Pseudomonas aeruginosa Modulates the Antiviral Response of Bronchial Epithelial Cells. Front. Immunol. 11:96. doi: 10.3389/fimmu.2020.00096
Boutin S, Weitnauer M, Hassel S, Graeber SY, Stahl M, Dittrich AS, Mall MA, Dalpke AH (2018): One time quantitative PCR detection of Pseudomonas aeruginosa to discriminate intermittent from chronic infection in cystic fibrosis. J Cyst Fibros. 17(3):348-355
Boutin S, Dalpke AH (2017): Acquisition and Adaptation of the airway microbiota in the early life of cystic fibrosis patients. Mol Cell Pediatr 4(1): 1-9. doi: 10.1186/s40348-016-0067-1.
Boutin S, Graeber SY, Stahl M, Dittrich SA, Mall MA and Dalpke AH (2017): Chronic but not intermittent infection with Pseudomonas aeruginosa is associated with global changes of the lung microbiome in cystic fibrosis. Eur Respir J 50 (4): 1701086, doi: 10.1183/13993003.01086-2017
Supported by Mukoviszidose e.V.
Links:
Curriculum Vitae
| Since 2022 | Director, Medical Microbiology and Hygiene, Dept. of Infectious Diseases, Med. Faculty, University Hospital Heidelberg |
| Since 2021 | PI, TRR319 “RMaP, RNA modifications and processing”, project A3 |
| 2019-2022 | Director, Institute of Med. Microbiology and Hygiene, Institute of Virology, Med. Faculty, Technical University Dresden |
| 2015-2018 | Chairman of the Habilitation Committee I, Medical Faculty Heidelberg |
| 2014-2018 | PI, German Center for Lung Research (DZL), TLRC Heidelberg |
| 2012-2015 | Coordinator Teaching & Education Activities, DZIF, Heidelberg site |
| 2011 | Consultant in microbiology, virology and epidemiology of infections |
| 2011-2015 | Member of SFB 938 “Environment specific control of immunological reactivity"; project E |
| 2008-2011 | Speaker of the Postgraduate Program "Differential activation and integration of signaling modules within the immune system"; postgraduate program of Baden-Württemberg |
| 2007-2018 | Member of the "Hartmut Hoffmann-Berling International Graduate School of Molecular and Cellular Biology" |
| 2006-2009 | Member of the SFB405 "Immunotolerance and its disturbances"; project B18 |
| 2006-2018 | Professor (W3) for Medical Microbiology and Infection and Immunity, Dept. of Medical Microbiology and Hygiene, University Heidelberg |
| 2006 | Specialist in immunology ("Fachimmunologe DGfl", German Society for Immunology |
| 2005-2006 | Group Leader, Dept. of Medical Microbiology and Hygiene, University Heidelberg |
| 2004-2008 | Member of the DFG priority programm SP1110 "Innate Immunity" |
| 2004 | Habilitation, university lecturer for infection and immunity Topic: Suppressor of Cytokine Signaling (SOCS) proteins as regulators of the activity of the innate immune system |
| 1999-2004 | Post-doc and Research Assistant, Inst. of Medical Microbiology, Philipps-University Marburg |
| 1998-1999 | First-year resident, Kreis- und Stadtkrankenhaus Alfeld, Internal medicine, License to practice medicine („Approbation als Arzt“) |
| 1998 | Graduation (MD), Med. Microbiology, Center of Hygiene and Human Genetics, University Göttingen, summa cum laude Topic: Manganese-Superoxide-Dismutase as a target of Epstein-Barr-Virus-induced autoantibodies and their impact on the pathogenesis of infectious mononucleosis |
| 1992-1998 | Medical student, Georg-August University Göttingen |
Lab members
My group just recently moved to Heidelberg. Positions available and applications welcome!
| Name | Function | Phone +49 6221 56- |
| Dr. Lan-Sun Chen | postdoc | 34783 |
| Andrew Tony-Odigie | postdoc | 310172 |
| Ivaneiá Valeriano-Nunes | PhD Student | |
| Luisa Breitenbach | master of science | |
| Paulina Schad | master of science | |
| Anette Fautsch | technician | 310065 |