Information for sponsors
Our Early Clinical Pharmacological Trial Unit KliPS is certified according to DIN EN ISO 9001:2015 and conducts trials of the early phases (Phase 0/I/II), including first-in-human trials as a certified ECTU.
Through standardized, GCP-compliant trial conduct and a highly qualified team with years of experience we ensure the greatest possible safety for trial participants and generate study data of the highest quality for industry partners and in academic research projects.
Our facility allows sponsors to conduct complex trials that require the expertise and capacities of a University Center. We recruit patients in collaboration with internal cooperation partners of the University Hospital and healthy volunteers in the vicinity of the research-oriented Heidelberg Campus and the entire Rhine-Neckar region. When conducting trials for sponsors, we benefit from our rich experience in planning and conducting self-initiated trials.
If you have any questions, please do not hesitate to contact us!
Your KLiPS ECTU-Team
Experience and infrastructure
| TRIAL PHASES |
|
|---|---|
| THERAPEUTICA |
|
| INDICATIONS |
|
| KEY POINTS |
|
| STUDY UNIT |
|
More information
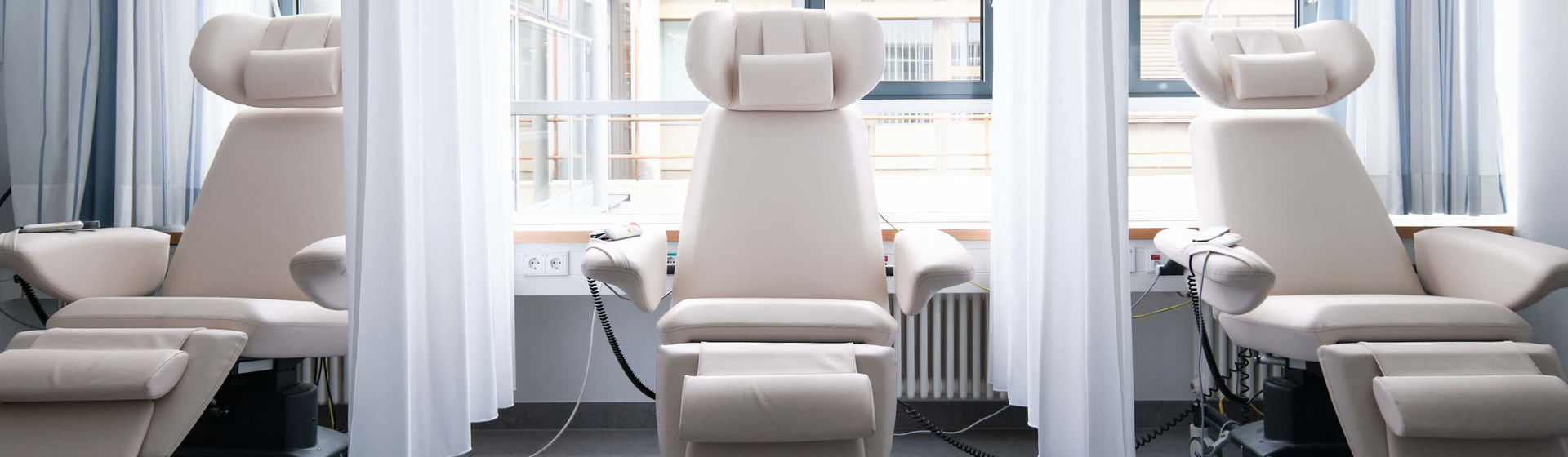
Infrastructure of our eCTU in the heart of the Medical Clinic
- Treatment in an inpatient day hospital or outpatient setting
- Equipment: 12 beds, 3 day hospital places, 2 outpatient rooms
- Central monitoring system (intermediate care setting)
- Access-protected area
- Temperature-controlled storage of medication and sample material
- Possibility to prepare biomarker samples in the Molecular Biology-Biochemistry Laboratory
- Comprehensive diagnostic possibilities of a maximum care university center
- Connection to the emergency and intensive care structures of the Medical Clinic
Our facility allows sponsors to conduct complex trials that require the expertise and capabilities of a University Center. We recruit patients in cooperation with internal partners of the University Hospital and healthy volunteers from the Heidelberg Campus and the entire Rhine-Neckar region. When conducting studies for sponsors, we benefit from our rich experience in planning and conducting self-initiated trials.
Main focus
- First-in-use trials in healthy volunteers and patients
- Dose-finding trials (phase II) in patients or volunteers (e. g. vaccination studies)
- Translational projects of the Heidelberg Campus
- Microdose trials with accompanying ultrasensitive drug analysis
- Pharmacokinetics (incl. frequent sampling)
- Drug interaction studies
- Genetic peculiarities in drug metabolism
- Trials concerning elimination disorders (liver cirrhosis, renal insufficiency, dialysis)
- Biomarker trials
Trial participant recruitment on the Heidelberg campus
Trial participant recruitment on the Heidelberg Campus
KliPS has ideal conditions for the successful recruitment of both healthy volunteers and patients for clinical trials. Through cooperation with the other clinical departments, there is access to large (partly rare) patient and subject cohorts with
- solid tumors (melanoma, gastrointestinal tumors, mamma-Ca) hematological diseases (e.g. multiple myeloma)
- infectious diseases (e.g. hepatitis B/D, herpes simplex virus)
- immunological diseases (e.g. psoriasis, Crohn's disease)
- cardiovascular diseases (e.g. dyslipidemias, amyloidosis, arrhythmias)
- elimination disorders (renal insufficiency incl. dialysis, liver cirrhosis)
- as well as to healthy volunteers without concomitant medication in different age categories (many of them genotyped for CYP and transporter polymorphisms).
Current cooperation partners
We cooperate with the following institutions and departments of Heidelberg University Hospital, among others
- Clinic for Hematology, Oncology and Rheumatology
- National Center for Tumor Diseases (NCT)
- Clinic for Gastroenterology, Infectious Diseases and Poisoning
- Clinic for Cardiology, Angiology and Pneumology
- German Cancer Research Center (DKFZ) Heidelberg Kidney Center
Information on the Network of Clinical Pharmacological Trial Centers KliPSnet
In order to cover the broadest possible spectrum of indications and to make optimal use of the existing possibilities at the Heidelberg Campus for the benefit of patients, KliPSnet, a close association of several clinical trial centers, has been founded in 2019. The goal of these centers is the joint planning, implementation and evaluation of sophisticated drug trials in the early clinical phases. They are united by common high quality standards, which is also expressed in the joint certification according to the quality management norm DIN EN ISO 9001:2015.
At present, in addition to the Early Clinical Pharmacological Trial Unit (KliPS), the network consists of the Pediatric Clinical Pharmacological Trial Center paedKliPS and the Pneumological Clinical Pharmacological Trial Center ThoraxKliPS, which specialize in studies with children and adolescents and studies with pneumological indications (incl. Medical Devices Trials), respectively.
![[Translate to English:] [Translate to English:]](/fileadmin/_processed_/5/6/csm_willkommen_d34eda0786.png)

![[Translate to English:] [Translate to English:]](/fileadmin/_processed_/8/5/csm_ACL_11_GrafikFingerTablet_LB8_c4ce258375.jpg)
![[Translate to English:] [Translate to English:]](/fileadmin/_processed_/3/a/csm_KS_b1_PatBett_Banner_719c5df562.jpg)
![[Translate to English:] [Translate to English:]](/fileadmin/_processed_/6/4/csm_publikaitonen_4abdce50fc.png)




